Dr. khaled al zubi lecture
for slides of Dr. Khaled alZubi lecture slides
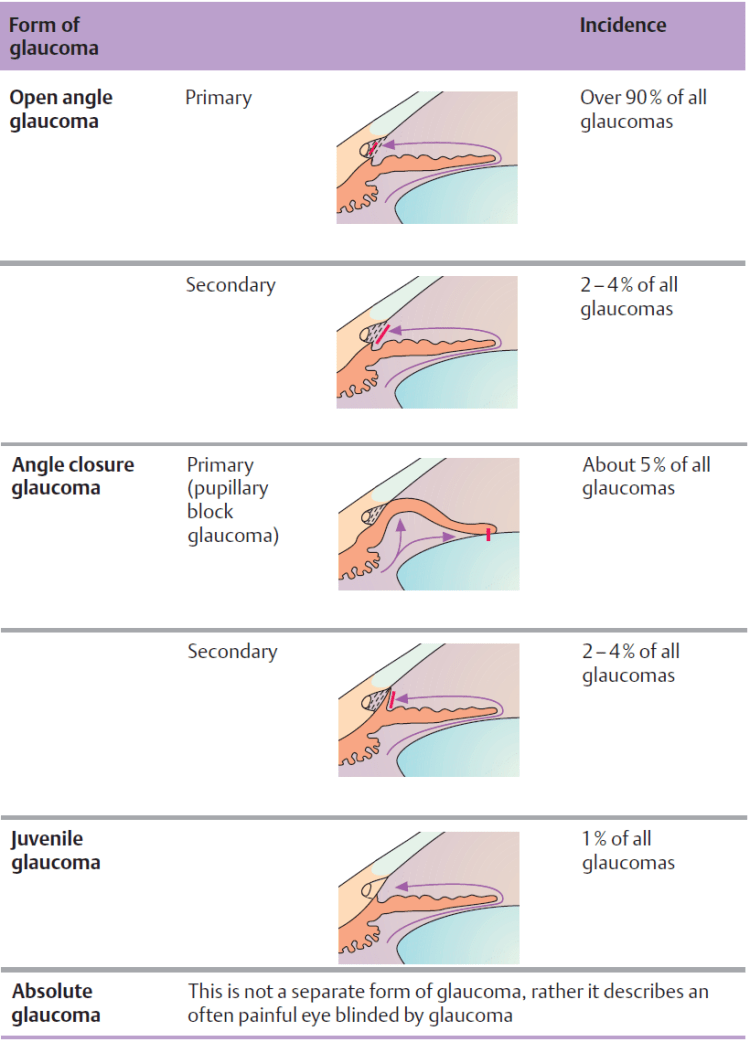
Definition: Glaucoma is a disorder in which increased intraocular pressure damages the optic nerve. This eventually leads to blindness in the affected eye.
Primary glaucoma refers to glaucoma that is not caused by other ocular disorders.
Secondary glaucoma may occur as the result of another ocular disorder or an undesired side effect of medication or other therapy.
Epidemiology: Glaucoma is the second most frequent cause of blindness in developing countries after diabetes mellitus. Fifteen to twenty per cent of all blind persons lost their eyesight as a result of glaucoma. In Germany, approximately 10% of the population over 40 has increased intraocular pressure.
Approximately 10% of patients seen by ophthalmologists suffer from glaucoma. Of the German population, 8 million persons are at risk of developing glaucoma, 800 000 have already developed the disease (i.e., they have glaucoma that has been diagnosed by an ophthalmologist), and 80 000 face the risk of going blind if the glaucoma is not diagnosed and treated in time.
Early detection of glaucoma is one of the highest priorities for the public health system.
Physiology and pathophysiology of aqueous humor circulation, The average normal intraocular pressure of 15mm Hg in adults is significantly higher than the average tissue pressure in almost every other organ in the body. Such a high pressure is important for the optical imaging and helps to ensure several things:
1- Uniformly smooth curvature of the surface of the cornea.
2- Constant distance between the cornea, lens, and retina.
3- Uniform alignment of the photoreceptors of the retina and the pigmented epithelium on Bruch’s membrane, which is normally taut and smooth. The aqueous humor is formed by the ciliary processes and secreted into the posterior chamber of the eye. At a rate of about 2–6 μl per minute and a total anterior and posterior chamber volume of about 0.2–0.4ml, about 1–2% of the aqueous humor is replaced each minute. The aqueous humor passes through the pupil into the anterior chamber. As the iris lies flat along the anterior surface of the lens, the aqueous humor cannot overcome this pupillary resistance (first physiologic resistance) until sufficient pressure has built up to lift the iris off the surface of the lens. Therefore, the flow of the aqueous humor from the posterior chamber into the anterior chamber is not continuous but pulsatile.

picture showing the circulation of aqueous inside the eye. A- is the non pigmented ciliary processes from were the aqueos is formed in an ATP dependent mechanism. B posterior chamber through the pupil to the trabecular mesh-work in C . which is will drain to the episcleral blood vessels ( this is called conventional pathway). pathway E which is the alternative pathway, filtration of the aqueous through the cilliary body.
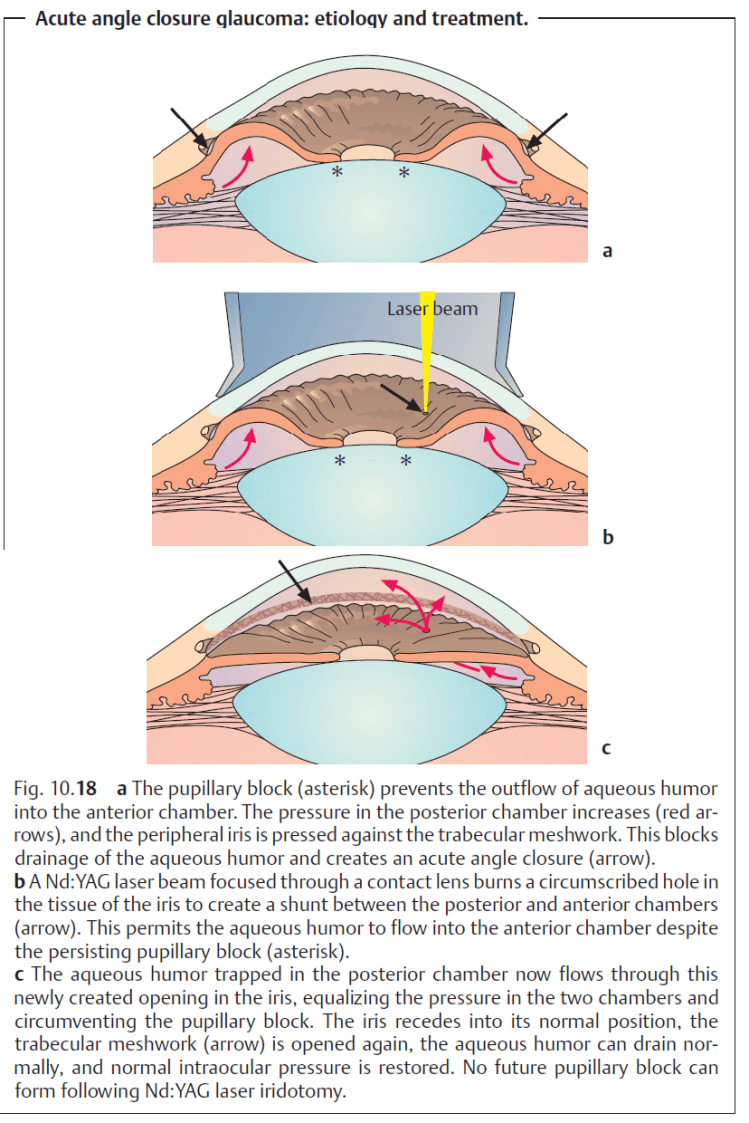
Any increase in the resistance to pupillary outflow (pupillary block) leads to an increase in the pressure in the posterior chamber; the iris inflates anteriorly on its root like a sail and presses against the trabecular meshwork. This is the pathogenesis of angle closure glaucoma.
Various factors can increase the resistance to pupillary outflow (Table10.1). The aqueous humor flows out of the angle of the anterior chamber through two channels:
- The trabecular meshwork receives about 85% of the outflow, which then drains into the canal of Schlemm. From here it is conducted by 20–30 radial collecting channels into the episcleral venous plexus (D).
- A uveoscleral vascular system receives about 15% of the outflow, which
joins the venous blood (E).
The trabecular meshwork (C) is the second source of physiologic resistance. The trabecular meshwork is a body of loose sponge-like avascular tissue between the scleral spur and Schwalbe’s line. Increased resistance in present in open angle glaucoma.
Classification: Glaucoma can be classified according to the specific pathophysiology (Table bellow). The many various types of glaucoma are nearly all attributable to
increased resistance to outflow and not to heightened secretion of aqueous humor.

Examination Methods
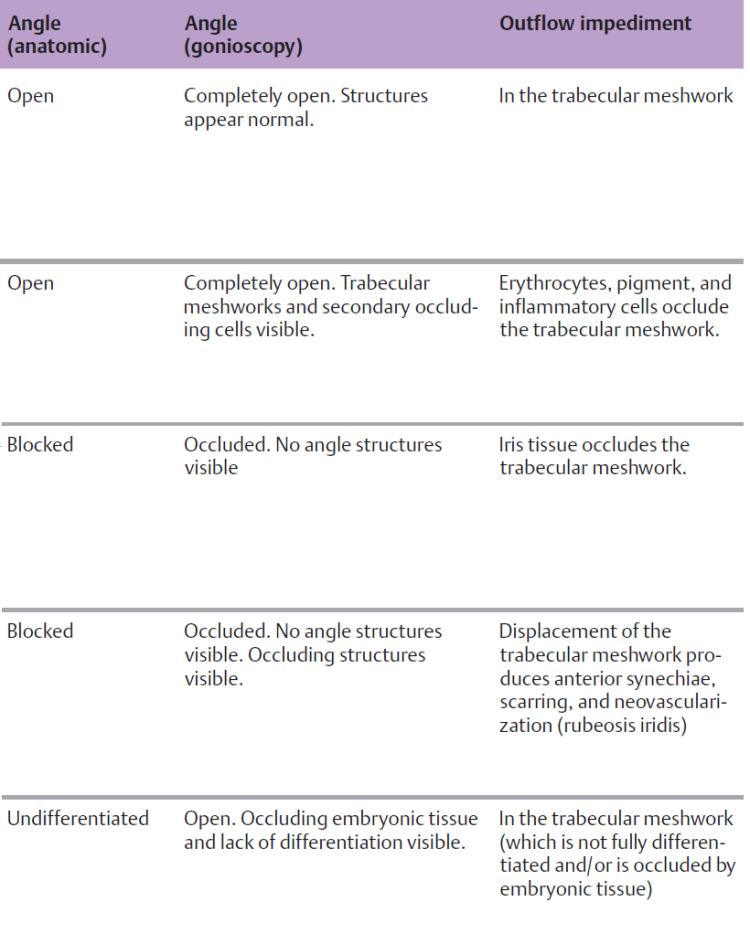
Gonioscopy is essential for further evaluation. To evaluate the depth of the anterior chamber with a slit-lamp biomicroscope, select a narrow setting for the light beam. The beam should strike the eye at a slight angle to the examiner’s line of sight.
Gonioscopy
The angle of the anterior chamber is evaluated with a gonioscope placed directly on the cornea.

Gonioscopy can differentiate the following conditions:
- Open angle: open angle glaucoma.
- Occluded angle: angle closure glaucoma.
- Angle access is narrowed: configuration with imminent risk angle of an acute closure glaucoma.
- Angle is occluded: secondary angle closure glaucoma, for example due to neovascularization in rubeosis iridis.
- Angle open but with inflammatory cellular deposits, erythrocytes, or pigment in the trabecular meshwork: secondary open angle glaucoma.
Gonioscopy is the examination of choice for identifying the respective presenting form of glaucoma.
Measuring Intraocular Pressure
- Palpation: Comparative palpation of both eyeballs is a preliminary examination that can detect increased intraocular pressure.
- If the examiner can indent the eyeball, which fluctuates under palpation, pressure is less than 20mm Hg.
- An eyeball that is not resilient but rock hard is a sign of about 60–70mm Hg of pressure (acute angle closure glaucoma).
- Schiøtz indentation tonometry: This examination measures the degree to which the cornea can be indented in the supine patient. The lower the intraocular pressure, the deeper the tonometer pin sinks and the greater distance the needle moves. Indentation tonometry often provides inexact results. For example the rigidity of the sclera is reduced inmyopic eyes, which will cause the tonometer pin to sink more deeply for that reason alone. Because of this, indentation tonometry has been largely supplanted by applanation tonometry.
- Applanation tonometry: This method is the most common method of measuring intraocular pressure. It permits the examiner to obtain a measurement on a sitting patient within a few seconds (Goldmann’s method) or on a supine patient (Draeger’s method). A flat tonometer tip has a diameter of 3.06mm for applanation of the cornea over a corresponding area (7.35 mm2). This method eliminates the rigidity of the sclera as a source of error (see also tonometric self-examination). Intraocular pressure of 22 mm Hg is regarded as suspicious.
Advantages:
- Does not require the use of a topical anesthetic.
- Non-contact measurement eliminates risk of infection (may be used to measure intraocular pressure in the presence of conjunctivitis).
Disadvantages:
- Calibration is difficult.
- Precise measurements are possible only within low to middle range pressures.
- Cannot be used in the presence of corneal scarring.
- Examination is unpleasant for the patient.
- Air flow is loud.
- The instrument is more expensive to purchase than an applanation tonometer.
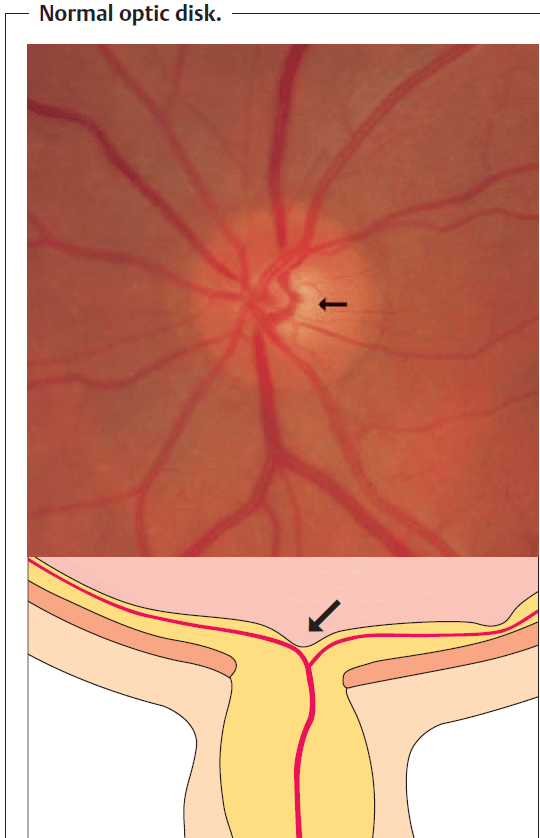
Optic Disk Ophthalmoscopy
The optic disk has a physiologic indentation known as the optic cup. In the presence of persistently elevated intraocular pressure, the optic cup becomes enlarged and can be evaluated by ophthalmoscopy.
Stereoscopic examination of the optic disk through a slit-lamp biomicroscope fitted with a contact lens provides a three-dimensional image. The optic cup may be examined stereoscopically with the pupil dilated.
The optic nerve is the eye’s “glaucoma memory.” Evaluating this structure will tell the examiner whether damage from glaucoma is present and how far advanced it is.
Normal optic cup: The normal anatomy can vary widely. Large normal optic cups are nearly always round and differ from the vertical elongation of the optic cup seen in eyes with glaucoma.
Documenting the optic disk: Recording findings in sketches is suitable for routine documentation and follow-up examination of the optic disk. Photographing the optic disk with a fundus camera permits longer-term followup.
Stereoscopic photography also provides a three-dimensional image. Optic disk measurement and tomography can provide precise measurements of the optic nerve.
Optic disk measurement. The area of the optic disk, optic cup, and neuroretinal rim (vital optic disk tissue) can be measured by planimetry on two-dimensional photographs of the optic nerve.
Optic disk tomography. Modern laser scanning ophthalmoscopes permit three-dimensional documentation of the optic nerve.
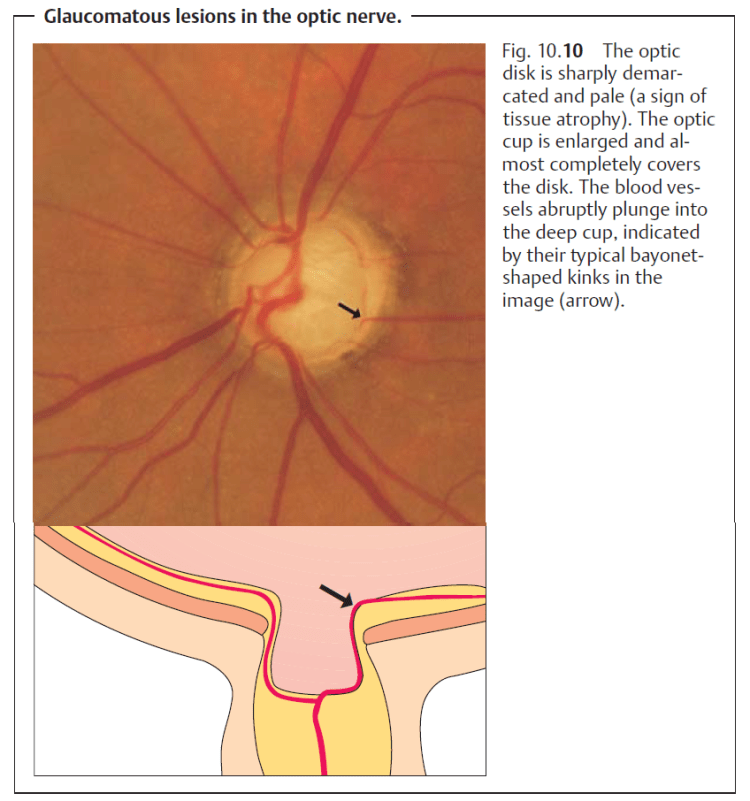
Glaucomatous changes in the optic nerve: Glaucoma produces typical changes in the shape of the optic cup. Progressive destruction of nerve fibers, fibrous and vascular tissue, and glial tissue will be observable. This tissue atrophy leads to an increase in the size of the optic cup and to pale discoloration of the optic disk. Progressive glaucomatous changes in the optic disk are closely associated with increasing visual field defects.
Visual Field Testing
Detecting glaucoma as early as possible requires documenting glaucomatous visual field defects at the earliest possible stage.We know that glaucomatous visual field defects initially manifest themselves in the superior paracentral nasal visual field or, less frequently, in the inferior field, as relative scotomas that later progress to absolute scotomas.
Computerized static perimetry (measurement of the sensitivity to differences in light) is superior to any kinetic method in detecting these early glaucomatous visual field defects. Computer-controlled semiautomatic grid perimetry devices such as the Octopus or Humphrey field analyzer are used to examine the central 30 degree field of vision (modern campimetry.
Reproducible visual field findings are important in follow-up to exclude any enlargement of the defects.

Primary Glaucoma
Primary Open Angle Glaucoma
Definition
Primary open angle glaucoma begins in middle-aged and elderly patients with minimal symptoms that progressively worsen. The angle of the anterior chamber characteristically remains open throughout the clinical course of the disorder. Epidemiology: Primary open angle glaucoma is by far themost common form of glaucoma and accounts for over 90% of adult glaucomas. The incidence of the disorder significantly increases beyond the age of 40, reaching a peak between the ages of 60 and 70. Its prevalence among 40-year-olds is 0.9% as compared to 4.7% among patients over the age of 50.
There appears to be a genetic predisposition for primary open angle glaucoma. Over one- third of glaucoma patients have relatives with the same disorder. Patients with a positive family history are at greater risk of developing the disorder.
Etiology: The cause of primary open angle glaucoma is not known, although it is known that drainage of the aqueous humor is impeded. The primary lesion occurs in the neuroretinal tissue of the optic nerve as compression neuropathy of the optic nerve.
Symptoms: The majority of patients with primary open angle glaucoma do not experience any subjective symptoms for years. However, a small number of patients experience occasional unspecific symptoms such as headache, a burning sensation in the eyes, or blurred or decreased vision that the patient may attribute to lack of eyeglasses or insufficient correction. The patient may also perceive rings of color around light sources at night, which has traditionally been regarded as a symptom of angle closure glaucoma.
Primary open angle glaucoma often does not exhibit typical symptoms for years. Regular examination by an ophthalmologist is crucial for early diagnosis.
Primary open angle glaucoma can be far advanced before the patient notices an extensive visual field defect in one or both eyes.
It is crucial to diagnose the disorder as early as possible because the prognosis for glaucoma detected in its early stages is far better than for advanced glaucoma. Where increased intraocular pressure remains undiagnosed or untreated for years, glaucomatous optic nerve damage and the associated visual field defect will increase to the point of blindness.
Diagnostic considerations: Measurement of intraocular pressure. Elevated intraocular pressure in a routine ophthalmic examination is an alarming sign. Twenty-four-hour pressure curve. Fluctuations in intraocular pressure of over 5–6mm Hg may occur over a 24-hour period. Gonioscopy. The angle of the anterior chamber is open and appears as normal as the angle in patients without glaucoma. Ophthalmoscopy. Examination of the optic nerve reveals whether glaucomatous cupping has already occurred and how far advanced the glaucoma is. Where the optic disk and visual field are normal, ophthalmoscopic examination of the posterior pole under green light may reveal fascicular nerve fiber defects as early abnormal findings.
Differential diagnosis:
Two disorders are important in this context:
- Ocular hypertension. Patients with ocular hypertension have significantly increased intraocular pressure over a period of yearswithout signs of glaucomatous optic nerve damage or visual field defects. Some patients in this group will continue to have elevated intraocular pressure but will not develop glaucomatous lesions; the otherswill develop primary open angle glaucoma. The probability that a patient will develop definitive glaucoma increases the higher the intraocular pressure, the younger the patient, and the more compelling the evidence of a history of glaucoma in the family.
- Low-tension glaucoma. Patients with low-tension glaucoma exhibit typical
progressive glaucomatous changes in the optic disk and visual field without
elevated intraocular pressure. These patients are very difficult to treat
because management cannot focus on the control of intraocular pressure.
Often these patients will have a history of hemodynamic crises such as
gastrointestinal or uterine bleeding with significant loss of blood, low blood
pressure, and peripheral vascular spasms (cold hands and feet). Patients with
glaucoma may also experience further worsening of the visual field due to a
drop in blood pressure. - Caution should be exercised when using cardiovascular and anti-hypertension
medications in patients with glaucoma. - Treatment: Indications for initiating treatment.
- Glaucomatous changes in the optic cup: Medical treatment should be initiated where there are signs of glaucomatous changes in the optic cup or where there is a difference of more than 20% between the optic cups of the two eyes.
- Any intraocular pressure exceeding 30mm Hg should be treated
- Increasing glaucomatous changes in the optic cup or increasing visual field defects: Regardless of the pressure measured, these changes showthat the current pressure level is too high for the optic nerve and that additional medical therapy is indicated. This also applies to patients with advanced glaucomatous damage and threshold pressure levels (around 22mm Hg). The strongest possible medications are indicated in these cases to lower pressure as much as possible (10–12mm Hg).
- Early stages: It is often difficult to determine whether therapy is indicated in the early stages, especially where intraocular pressure is elevated slightly above threshold values. Patients with low-tension glaucoma exhibit increasing cupping of the optical disk even at normal pressures (less than 22mmHg), whereas patients with elevated intraocular pressure (25–33mm Hg) may exhibit an unchanged optic nerve for years. Patients with suspected glaucoma and risk factors such as a family history of the disorder, middle myopia, glaucoma in the other eye, or differences
between the optic cup in the two eyes should be monitored closely. Follow-up
examinations should be performed three to four times a year, especially for
patients not undergoing treatment.
Medical therapy. Available options in medical treatment of glaucoma:
- Inhibit aqueous humor production.
- Increase trabecular outflow.
- Increase uveoscleral outflow.

- Principles of medical treatment of primary open angle glaucoma:
- Medical therapy is the treatment of choice for primary open angle glaucoma.
- Surgery is indicated only where medical therapy fails.
There is no one generally applicable therapy plan. However, several principles may be formulated:
- Where miosis is undesirable, therapy should begin with beta blockers.
- Where miosis is not a problem (as is the case with aphakia), therapy begins with miotic agents.
- Miotic agents may be supplemented with beta blockers, epinephrine derivatives, guanethidine, dorzolamide and/or latanoprost maximum topical therapy). Osmotic agents or carbonic anhydrase inhibitors (administered orally or intravenously) inhibit the production of aqueous humor. They can be administered temporarily in addition to topical medications. Their side effects usually make them unsuitable for prolonged treatment. The general rule is to try to use the weakest possible medications required to achieve normal pressure over a 24-hour period: as much as necessary, and as little as possible.
- The effectiveness of any pressure-reducing therapy should be verified by pressure analysis on the ward or on an outpatient basis.
The effect of the eye drops should not interfere with the patient’s ability to work. Tolerance, effects, and side effects of the eyedrops should be repeatedly verified on an individual basis during the course of treatment.
- Surgical treatment of primary open angle glaucoma.
Indications:
- Medical therapy is insufficient.
- The patient does not tolerate medical therapy. Reactions include allergy, reduced vision due to narrowing of the pupil, pain, and ciliary spasms, and ptosis.
- The patient is not a suitable candidate for medical therapy due to lack of compliance or dexterity in applying eye drops.
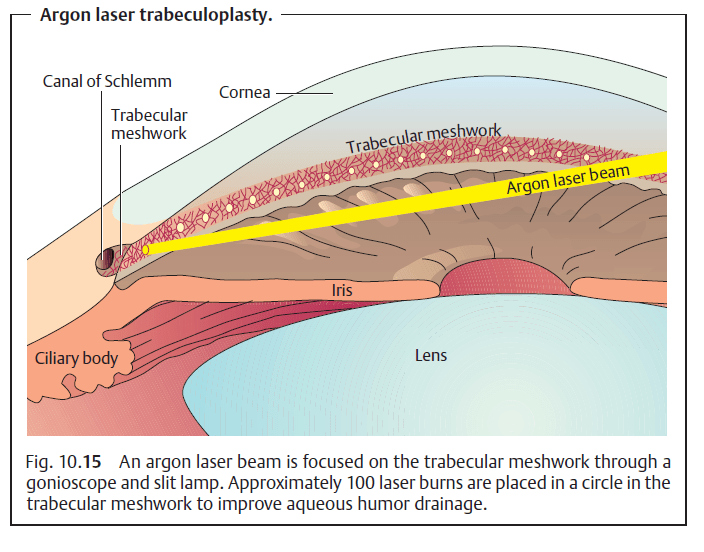
- Argon laser trabeculoplasty:
Principle: Laser burns in the trabecular meshwork cause tissue contractionthat widens the intervening spaces and improves outflowthrough the trabecular meshwork.
- Filtration surgery:
Principle: The aqueous humor is drained through the anterior chamber through a subconjunctival scleral opening, circumventing the trabecular meshwork. Formation of a thin-walled filtration bleb is a sign of sufficient drainage of aqueous humor. - Filtration surgery:
Principle: The aqueous humor is drained through the anterior chamber through a subconjunctival scleral opening, circumventing the trabecular meshwork. Formation of a thin-walled filtration bleb is a sign of sufficient drainage of aqueous humor. - Closed angle glaucoma treatment: Peripheral iridectomy (incisional procedure): Where the cornea is still swollen with edema or the iris is very thick, an open procedure may be required to create a shunt. A limbal incision is made at 12 o’clock under topical anesthesia or general anesthesia, through which a basal iridectomy is performed. Today peripheral iridectomy is rarely performed, in only in 1–2% of all cases.
Secondary Glaucomas
Definition: These glaucomas are caused by other ocular diseases of factors such as inflammation, trauma, bleeding, tumors, medication, and physical or chemical influences.
- The most important forms: Pseudoexfoliative glaucoma. This form occurs particularly frequently in Scandinavian countries. Deposits of amorphous acellular material form throughout the anterior chamber and congest the trabecular meshwork.
- Pigmentary glaucoma. Young myopic men typically are affected. The disorder is characterized by release of pigment granules from the pigmentary epithelium of the iris that congest the trabecular meshwork. Cortisone glaucoma. Thirty-five to forty per cent of the population react to three-week topical or systemic steroid therapy with elevated intraocular pressure. Increased deposits of mucopolysaccharides in the trabecular meshwork presumably increase resistance to outflow; this is reversible when the steroids are discontinued.
- Inflammatory glaucoma. Two mechanisms contribute to the increase in intraocular pressure:
1. The viscosity of the aqueous humor increases as a result of the influx of protein from inflamed iris vessels.
2. The trabecular meshwork becomes congested with inflammatory cells and
cellular debris.
- Phacolytic glaucoma. This is acute glaucoma in eyes with mature or hypermature cataracts. Denatured lens protein passes through the intact lens capsule into the anterior chamber and is phagocytized. The trabecular meshwork becomes congestedwith protein-binding macrophages and the protein itself.
Secondary Angle Closure Glaucoma
Definition: In secondary angle closure glaucoma as in primary angle closure glaucoma, the increase in intraocular pressure is due to blockage of the trabecular meshwork. However, the primary configuration of the anterior chamber is not the decisive factor.
The most important causes: Rubeosis iridis. Neovascularization draws the angle of the anterior chamber together like a zipper (neovascular glaucoma). Ischemic retinal disorders such as diabetic retinopathy and retinal vein occlusion can lead to rubeosis iridis with progressive closure of the angle of the anterior chamber.Other forms of retinopathy or intraocular tumors can also cause rubeosis iridis. The prognosis for eyes with neovascular glaucoma is poor.
Trauma. Post-traumatic presence of blood or exudate in the angle of the anterior chamber and prolonged contact between the iris and trabecular meshwork in a collapsed anterior chamber (following injury, surgery, or insufficient treatment of primary angle closure) can lead to anterior synechiae and angle closure without rubeosis iridis.

Treatment of secondary glaucomas:
Medical therapy of secondary glaucomas is usually identical to the treatment of primary chronic open angle glaucoma. Secondary glaucomas may be caused by many different factors, and the angle may be open or closed. Therefore, treatment will depend on the etiology of the glaucoma. The underlying disorder is best treated first. Glaucomas with uveitis (such as iritis or iridocyclitis) initially are treated conservatively with anti-inflammatory and antiglaucoma agents. Surgery is indicated where conservative treatment is not sufficient. The prognosis for secondary glaucomas is generally worse than for primary glaucomas.
Childhood Glaucomas
Definition: Any abnormal increase in intraocular pressure during the first years of life will cause dilatation of the wall of the globe, and especially of the cornea. The result is a characteristic, abnormally large eye (buphthalmos) with a progressive increase in corneal diameter. This is also referred to as hydrophthalmos or hydrophthalmia.
Epidemiology: Glaucomas in children occur once every 12000–18000 births and account for about 1% of all glaucomas. Primary congenital glaucoma is an inherited autosomal recessive disorder. It is bilateral in approximately 70% of all cases; boys are affected in approximately 70% of all cases; and glaucoma manifests itself before the age of six months in approximately 70% of all cases. Today there is widespread public awareness of glaucoma in adults. Unfortunately, this does not yet apply to glaucoma in children.
- Symptoms: Classic signs include photophobia, epiphora, corneal opacification, and unilateral or bilateral enlargement of the cornea. These changes may
be present from birth (in congenital glaucoma) or may develop shortly after
birth or during the first few years of life. Children with this disorder are irritable, poor eaters, and rub their eyes often. The behavior of some children may lead one to suspect mental retardation. Physicians should be alert to parents who boast bout their child’s “big beautiful eyes” and should measure intraocular pressure. It is essential to diagnose the disorder as early in the child’s life as possible to minimize the risk of loss of or irreparable damage to the child’s vision.
Diagnostic considerations: These examinations may be performed without general anesthesia in many children. However, general anesthesia will occasionally be necessary to confirm the diagnosis especially in older children - Prognosis: Goniotomies and trabeculotomies are not always successful. Even after apparently successful initial trabecular surgery, these children require a lifetime of follow-up examinations (initially several times a year and later once every year) as elevated intraocular pressure can recur, in which case repeat goniotomy or trabeculotomy is indicated.
